How Glioblastoma Treatment Works: From Surgery to New Therapies
Dr. Arunav Sharma
Conventional advice often reduces glioblastoma care to a single pathway. That view is incomplete. In practice, How Glioblastoma Treatment Works: Chemotherapy to Surgery is a coordinated sequence that adapts to tumour biology, anatomy, and patient priorities. I will map that sequence clearly, from maximal safe resection to emerging cellular therapies, and I will show how choices link to outcomes. It is basically a system of trade-offs and timing. And yet, detail by detail, precision matters.
Current Standard Treatment Approaches for Glioblastoma
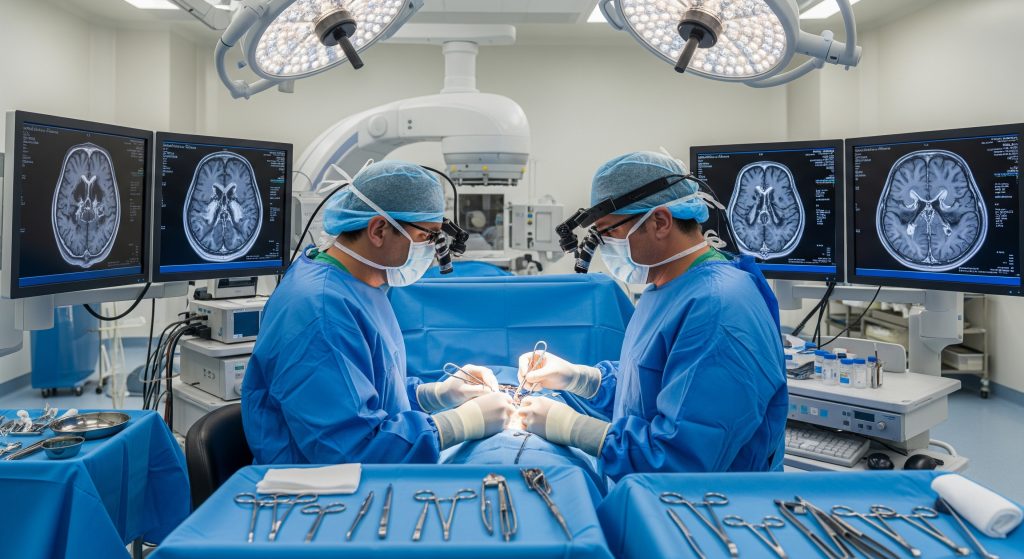
1. Surgical Resection: Maximal Safe Removal
I start with surgery because How Glioblastoma Treatment Works: Chemotherapy to Surgery is anchored by cytoreduction. Neurosurgical teams pursue maximal safe resection, balancing aggressive debulking with preservation of function. The aim is to remove enhancing tumour while respecting eloquent cortex and critical tracts.
-
Planning uses high-resolution MRI, tractography, and sometimes functional mapping.
-
Techniques include awake craniotomy, intraoperative MRI, and fluorescence guidance.
-
Outcome hinges on extent of resection, location, and performance status.
In practice, I look for a strategy that maximises volumetric reduction with minimal neurological deficit. A small anecdote illustrates the point. A left temporal lesion with language involvement may benefit from awake mapping and staged goals, rather than a single radical attempt. Precision over bravado. This is How Glioblastoma Treatment Works: Chemotherapy to Surgery when safety is non-negotiable.
2. Radiation Therapy: Standard Protocols
Postoperative radiotherapy consolidates local control. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, conventional external beam radiation remains the backbone after surgery. Fractionation typically uses daily small doses over several weeks, targeting the resection cavity with a margin to cover microscopic disease.
-
Target volumes are derived from preoperative and postoperative imaging.
-
Planning prioritises dose conformity and organ-at-risk constraints.
-
Concurrent temozolomide is standard for fit adults without conflicting contraindications.
Modern planning systems allow tighter fields, but biology still drives recurrence patterns. The reality is sobering. Marginal failure occurs, yet most regrowth remains near the original site. That is why I align expectations early and integrate supportive care from the start. It is a disciplined step in How Glioblastoma Treatment Works: Chemotherapy to Surgery.
3. Temozolomide Chemotherapy: Administration and Mechanism
Temozolomide is an oral alkylating agent. Within How Glioblastoma Treatment Works: Chemotherapy to Surgery, it acts as the radiosensitiser during radiotherapy and as maintenance therapy thereafter. The drug methylates DNA at specific positions, disrupting tumour replication and promoting cell death.
-
Administration follows a concurrent phase, then cycles of maintenance therapy.
-
Monitoring covers blood counts, hepatic profile, and symptom burden.
-
Support includes antiemetics and prophylaxis where clinically indicated.
Mechanistically, temozolomide exploits vulnerabilities in tumour DNA repair capacity. MGMT promoter methylation can influence sensitivity, which I discuss later. Do I see perfect responses? Rarely. Do I see meaningful disease control for many? Yes, particularly when the schedule is delivered with careful dose management. That steadiness reflects How Glioblastoma Treatment Works: Chemotherapy to Surgery in routine practice.
4. Combination Therapy: The Stupp Protocol
The Stupp approach formalised the concurrent and adjuvant sequence that many now call standard of care. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, this protocol ties surgery, radiation, and temozolomide into one coherent course.
-
Maximal safe resection or diagnostic biopsy.
-
Radiotherapy with concurrent temozolomide.
-
Adjuvant cycles of temozolomide with imaging surveillance.
Why does it endure? Because it balances efficacy, tolerability, and logistics. It also sets the platform for trial participation at recurrence. Critics argue the gains are modest. They are not entirely wrong, but they underplay the fact that consistency across centres enables predictable planning and timely escalation. That operational reliability is part of How Glioblastoma Treatment Works: Chemotherapy to Surgery.
Latest Treatment Innovations and Clinical Trials
1. Proton Beam Therapy with Advanced Imaging
Protons offer a sharper fall-off in dose beyond the target. I consider it when anatomy makes sparing healthy tissue vital. Within How Glioblastoma Treatment Works: Chemotherapy to Surgery, protons add value by reducing integral dose to normal brain while maintaining tumour coverage.
-
Advanced planning integrates diffusion imaging and functional mapping.
-
Adaptive replanning can adjust to cavity changes during therapy.
-
The clinical question is not only control, but also quality of life and cognitive preservation.
Proton therapy is not a universal upgrade. It is a contextual tool, especially near critical structures in younger or long-horizon patients. As current data suggests, benefit varies by case mix and planning quality. That nuance belongs in How Glioblastoma Treatment Works: Chemotherapy to Surgery when personalising radiotherapy.
2. Ultrasound-Mediated Drug Delivery
Focused ultrasound paired with microbubbles can transiently open the blood-brain barrier. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, this technique aims to increase intratumoural drug concentrations without raising systemic toxicity.
-
Procedure timing aligns with systemic drug administration.
-
Imaging confirms target localisation and safety windows.
-
Early studies explore tolerability and pharmacokinetics first, then efficacy signals.
Here is why it matters. Even effective molecules struggle to cross protective barriers. If permeability improves in a controlled fashion, standard agents and novel payloads may act more decisively. It is a promising bridge between pharmacology and physics, and it fits into How Glioblastoma Treatment Works: Chemotherapy to Surgery as a delivery upgrade, not a replacement.
3. Gene Therapy Approaches
Gene therapy explores vectors that deliver tumour-killing instructions or sensitise cells to prodrugs. I see three broad tactics in How Glioblastoma Treatment Works: Chemotherapy to Surgery.
-
Oncolytic vectors that preferentially replicate in tumour tissue.
-
Gene transfer that disrupts survival pathways or immune evasion.
-
Suicide gene systems activated by a follow-on agent.
These approaches remain largely within clinical trials. The central challenge is achieving sufficient distribution across infiltrative margins. Delivery routes include intratumoural injection and, in some studies, convection-enhanced delivery. It is early, but it adds a new logic to How Glioblastoma Treatment Works: Chemotherapy to Surgery by attacking the biology, not just the bulk.
4. Targeted Molecular Treatments
Targeted agents pursue actionable alterations such as receptor tyrosine kinase activation or pathway dysregulation. Selection depends on molecular profiling. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, these treatments sit alongside or after standard therapy, often within trials.
-
Case selection uses tumour sequencing and immunohistochemistry.
-
Decision-making weighs blood-brain barrier penetration and resistance patterns.
-
Endpoints consider radiographic response and symptom control, not only size metrics.
The variety of targets means no single agent dominates. Arguably, matched therapy will expand as profiling improves and drug design becomes more brain-permeable. This is a measured evolution inside How Glioblastoma Treatment Works: Chemotherapy to Surgery.
CAR-T Cell Immunotherapy Advances
1. Dual-Target CAR-T Approaches
Chimeric antigen receptor T cells are engineered to recognise tumour antigens. Dual-target constructs aim to reduce antigen escape. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, CAR-T offers a cellular tactic that complements, rather than replaces, local therapies.
-
Designs may combine two antigen-binding domains on one construct.
-
Rationale: heterogeneous tumours lose single targets under pressure.
-
Monitoring includes cytokine dynamics and neurotoxicity vigilance.
This is sophisticated immunology meeting surgical oncology. And yet, distribution inside complex brain tissue remains a practical hurdle. The field is adjusting by redesigning receptors and delivery routes. It is how How Glioblastoma Treatment Works: Chemotherapy to Surgery incorporates live therapies.
2. GD2-Targeted CAR-T Therapy
GD2 is an antigen explored in neuro-oncology. GD2-targeted CAR-T products are being tested for feasibility and biological signal. Within How Glioblastoma Treatment Works: Chemotherapy to Surgery, these products may be deployed in recurrent settings with close monitoring.
-
Preclinical rationale stems from tumour-associated expression patterns.
-
Clinical observation focuses on safety, trafficking, and early response markers.
-
Combination strategies with conditioning or checkpoint agents are under study.
Outcomes vary, as far as current data suggests. Some patients show transient reductions or stabilisation. Others demonstrate limited persistence. Iteration is rapid. That is a healthy sign for How Glioblastoma Treatment Works: Chemotherapy to Surgery when adapting immunotherapy.
3. Intraventricular Delivery Methods
Intraventricular or intracavitary infusion seeks better regional distribution. For CAR-T, proximity to cerebrospinal fluid pathways may aid coverage. Within How Glioblastoma Treatment Works: Chemotherapy to Surgery, route of administration is not a detail. It is strategy.
-
Catheter placement is planned with neurosurgical precision.
-
Dosing schedules escalate cautiously with real-time safety checks.
-
Pharmacodynamic sampling leverages cerebrospinal fluid where appropriate.
When the route aligns with disease geography, early signals improve to an extent. But safety windows are narrow. A pragmatic rhythm is essential: treat, assess, adjust. That cadence fits the broader logic of How Glioblastoma Treatment Works: Chemotherapy to Surgery.
4. Clinical Trial Outcomes and Responses
Trial readouts in this area emphasise safety, feasible dosing, and biological activity. Durable remissions are uncommon, though not absent. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, I interpret emerging CAR-T data through three lenses.
-
Safety profile: cytokine release risk, neurological symptoms, steroid exposure.
-
Depth and durability: transient responses versus sustained control.
-
Integration: sequencing with surgery, radiation, and systemic therapy.
Expect uneven progress. It is how science often advances in complex diseases. The direction is well chosen, even if the path is winding. And that, again, is How Glioblastoma Treatment Works: Chemotherapy to Surgery in a field learning quickly.
Survival Rates and Prognostic Factors
Age and Overall Health Impact
Age and baseline function shape outcomes. Rather than quoting exact figures, I advise framing glioblastoma survival rates as a spectrum influenced by biology, comorbidity, and treatment intensity. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, I weigh the following carefully.
-
KPS, the Karnofsky Performance Status, signals resilience for intensive regimens.
-
Frailty and organ reserve inform chemotherapy dosing and radiation planning.
-
Rehabilitation access and support networks affect recovery speed.
Older patients can still benefit meaningfully, particularly with tailored regimens and vigilant toxicity management. A precise plan often outperforms a maximal plan. That is the practical side of How Glioblastoma Treatment Works: Chemotherapy to Surgery.
MGMT Methylation Status
MGMT promoter methylation correlates with better sensitivity to temozolomide. I interpret it as a probabilistic guide, not a binary switch. Inside How Glioblastoma Treatment Works: Chemotherapy to Surgery, an MGMT-methylated profile often supports full engagement with concurrent and adjuvant temozolomide.
|
Marker |
Implication in practice |
|---|---|
|
MGMT methylated |
Higher likelihood of benefit from temozolomide-based regimens. |
|
MGMT unmethylated |
Benefit less predictable; consider trials or alternative strategies. |
Testing methods can vary, so I correlate results with the full clinical picture. It is one factor among many in How Glioblastoma Treatment Works: Chemotherapy to Surgery.
IDH Mutation Significance
IDH mutation is uncommon in primary glioblastoma but carries prognostic weight. The biology differs, and outcomes may be relatively more favourable. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, IDH status helps refine counselling and trial selection.
-
Confirm classification using integrated histopathology and molecular testing.
-
Assess the role of alkylating therapy in the overall plan.
-
Watch for distinct imaging and growth kinetics in follow-up.
Here is the editorial point. Labels guide, but they do not treat. The care plan still lives in day-to-day execution. That is the essence of How Glioblastoma Treatment Works: Chemotherapy to Surgery.
Treatment Response Predictors
I group predictors into baseline, on-treatment, and post-treatment domains. It keeps the logic clean and actionable. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, this structure helps me adjust quickly.
-
Baseline: molecular markers, extent of resection, and KPS.
-
On-treatment: haematological tolerance, radiotherapy adherence, and early imaging signals.
-
Post-treatment: pattern of recurrence, steroid dependence, and neurocognition.
Response is usually mixed. Some metrics improve while others deteriorate. I privilege functional outcomes alongside imaging. Patients live their days, not their scans. That stance shapes How Glioblastoma Treatment Works: Chemotherapy to Surgery.
Looking Ahead: Future of Glioblastoma Treatment
The future will be molecularly steered, delivery-optimised, and data-integrated. I expect multi-modal regimens to include targeted agents, refined radiation, and engineered cells. In How Glioblastoma Treatment Works: Chemotherapy to Surgery, three shifts stand out.
-
Profiling at diagnosis to inform trial matching from day one.
-
Adaptive radiotherapy that evolves with cavity dynamics and response signals.
-
Combinatorial immunotherapy that addresses heterogeneity and immune suppression.
Clinically, I anticipate more day-1 trial discussions, leaner steroid use, and smarter symptom tracking. Technically, I expect better blood-brain barrier modulation and safer local delivery. Strategically, I foresee longitudinal decision support that unites imaging, pathology, and real-world function. And yet, one principle will not change. High-quality basics will remain decisive in How Glioblastoma Treatment Works: Chemotherapy to Surgery.
Frequently Asked Questions
What are the median survival rates for glioblastoma patients receiving standard treatment?
Median outcomes are often discussed in ranges rather than absolutes. A typical course produces survival measured in months beyond a year in many cases, with variation by MGMT status, age, and performance. I frame glioblastoma survival rates as bands, not promises, because biology and fitness shift the centre of gravity. This framing aligns with How Glioblastoma Treatment Works: Chemotherapy to Surgery, which treats median figures as context, not destiny.
How does temozolomide chemotherapy cross the blood-brain barrier?
Temozolomide is a small, lipophilic molecule that reaches the central nervous system in therapeutically relevant concentrations. Once inside, it exerts alkylating effects on tumour DNA. From a process perspective, this is a rare example of a systemic agent with predictable brain penetration. It is a cornerstone in How Glioblastoma Treatment Works: Chemotherapy to Surgery because administration is reliable and monitoring is straightforward.
What makes CAR-T cell therapy promising for glioblastoma treatment?
CAR-T embodies personalised potency. Cells are engineered to recognise tumour antigens and can adapt their activity within the microenvironment. Promise comes from specificity and the possibility of memory. Limitations include antigen heterogeneity and trafficking barriers. Even so, when integrated thoughtfully with surgery and radiation, the approach strengthens How Glioblastoma Treatment Works: Chemotherapy to Surgery with a new mechanism.
Which molecular markers predict better treatment response in glioblastoma?
MGMT promoter methylation and IDH mutation are the two most referenced markers. They correlate with sensitivity to alkylating agents and overall prognosis. Additional markers and signatures continue to emerge, but validation is uneven. I use these markers to guide expectations and to shape trial choices. This is pragmatic precision within How Glioblastoma Treatment Works: Chemotherapy to Surgery.
What are the most common side effects of temozolomide treatment?
Common effects include myelosuppression, nausea, fatigue, and occasional hepatic disturbances. Prophylaxis and supportive care mitigate many issues, and dose adjustments preserve continuity. Practical monitoring and early intervention are vital. This is how I stabilise quality of life while sustaining efficacy in How Glioblastoma Treatment Works: Chemotherapy to Surgery.
How do proton beam therapy and standard radiation compare for glioblastoma?
Protons can reduce radiation dose to surrounding normal brain by virtue of their physical properties. Clinical benefits depend on anatomy, plan quality, and patient factors. Conventional radiation remains highly effective and widely available. The decision is not binary; it is contextual. I select the modality that best serves the intended balance of control and function. That selection sits squarely inside How Glioblastoma Treatment Works: Chemotherapy to Surgery.







 We do what's right for you...
We do what's right for you...



