Pleural Effusion Management: A Complete Overview
Dr. Kunal Luthra
Pleural effusion management has always been about draining fluid and hoping for the best. For decades, that was the standard approach. Today, that thinking is not just outdated – it is actively leading to unnecessary complications and repeat procedures. The shift towards targeted intervention based on effusion type, real-time imaging, and patient-centred decision-making has fundamentally changed outcomes. Understanding pleural effusion pathophysiology is no longer optional knowledge for the curious; it is the foundation upon which every treatment decision must rest. This overview breaks down the critical management strategies, diagnostic pathways, and treatment selections that separate effective pleural effusion management from outdated guesswork.
Initial Management Approaches for Pleural Effusion
Think of managing pleural effusion like navigating a river with multiple tributaries. The initial approach determines which path yields the best outcome. Getting this wrong means backtracking, delays, and frustrated patients.
1. Thoracentesis as First-Line Diagnostic and Therapeutic Intervention
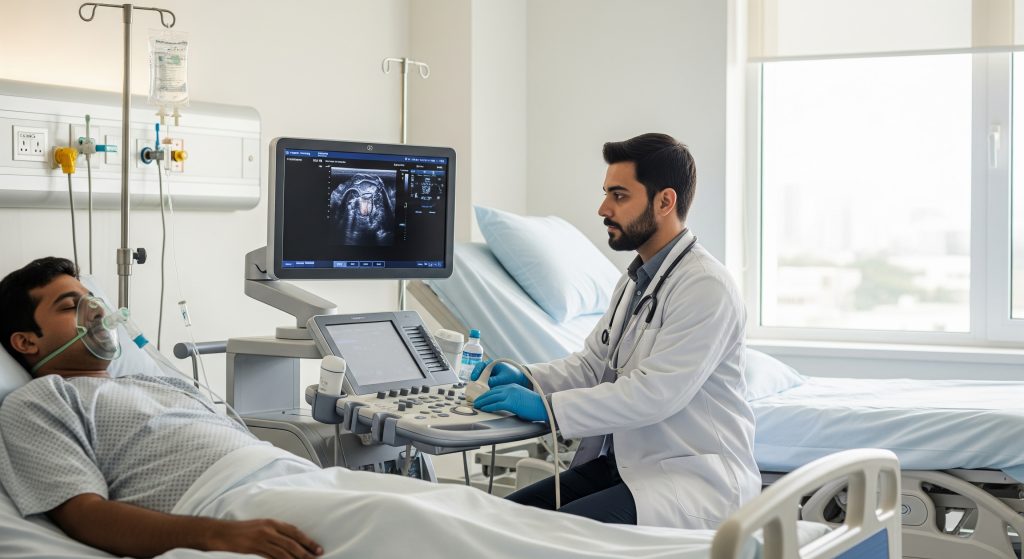
Thoracentesis remains the cornerstone of pleural effusion management. It is both diagnostic and therapeutic. A needle goes into the pleural space, fluid comes out, and suddenly there is clarity about what is happening inside the chest. The procedure provides immediate symptomatic relief for breathless patients while simultaneously yielding fluid for analysis.
The dual purpose makes it invaluable. Patients experiencing significant dyspnoea from large effusions often feel dramatic improvement within minutes of drainage. Meanwhile, the laboratory receives samples that can distinguish between benign and malignant processes, infectious causes, and systemic diseases.
But here is the catch. Thoracentesis performed blindly, without imaging guidance, carries real risks. Pneumothorax. Bleeding. Organ injury. The success rate drops and complication rates climb when clinicians rely solely on physical examination landmarks.
2. Ultrasound-Guided Procedures for Improved Safety
Ultrasound has transformed procedural safety in ways that textbooks from twenty years ago could never have predicted. According to Mayo Clinic, ultrasound guidance during thoracentesis reduces the risk of complications such as pneumothorax and bleeding, with ultrasound proving more sensitive than chest X-rays for detecting pleural fluid.
Point-of-care ultrasound allows real-time visualisation of the effusion, diaphragm position, and surrounding structures. The clinician can watch the needle enter the fluid collection. That visual confirmation changes everything. Research published in PMC confirms that point-of-care ultrasound can accurately evaluate and manage pleural effusions, enabling safe thoracentesis with real-time assessment.
In intensive care settings, where patients are often critically ill and cannot tolerate complications, this matters even more. Real-time ultrasound guidance during pleural effusion drainage significantly improves respiratory mechanics and reduces complications in ICU patients, as documented in studies from PMC.
Honestly, the only thing that really matters here is whether ultrasound is being used. Don’t even bother debating blind versus imaging-guided approaches. The evidence is overwhelming. Use ultrasound.
3. Chemical Pleurodesis with Talc
When pleural effusions keep coming back, pleurodesis becomes the conversation. Chemical pleurodesis creates adhesion between the parietal and visceral pleura using sclerosing agents. Talc is the workhorse here.
The mechanism is elegantly brutal. Talc induces a local inflammatory response, triggering mesothelial cells to secrete growth factors and cytokines. This activates the coagulation cascade and stimulates fibroblast proliferation, ultimately obliterating the pleural space. The pleura literally stick together, preventing fluid from reaccumulating.
For malignant pleural effusions specifically, talc pleurodesis delivers. Success rates hover around 84.6% within 30 days post-procedure, according to data from PubMed. That is a meaningful number for patients facing recurrent symptomatic effusions from cancer.
The procedure remains less invasive than surgical options while maintaining effectiveness. Factors influencing treatment choice include patient performance status, effusion type, and anticipated survival. A patient with a Karnofsky Performance Status suggesting limited life expectancy might benefit from pleurodesis to maximise quality of remaining time, while someone with better prognosis might warrant more aggressive approaches.
What drives me crazy is the delay in getting patients to pleurodesis. A retrospective analysis highlighted significant waiting times for procedures, hindering prompt alleviation of distressing symptoms. Patients are struggling to breathe while paperwork gets sorted. That is unacceptable.
4. Indwelling Pleural Catheters for Ambulatory Management
Not every patient is a candidate for pleurodesis. Some have non-expandable lungs that will not allow the pleural surfaces to appose. Others have limited life expectancy and want to spend it at home, not in hospital wards waiting for procedures.
Indwelling pleural catheters – IPCs – offer ambulatory management. A tunnelled catheter remains in place, allowing drainage at home. Patients or carers can drain fluid as symptoms dictate. This represents a fundamental shift from hospital-centric to patient-centric care.
The convenience is substantial. Regular drainage prevents the accumulation that causes dyspnoea. Some patients even achieve spontaneous pleurodesis over time through repeated drainage and inflammation.
5. Surgical Options Including VATS and Thoracoscopy
When medical management fails or diagnosis remains uncertain, surgical intervention enters the picture. Video-assisted thoracoscopic surgery (VATS) provides direct visualisation of the pleural space. Biopsies can be taken under vision. Loculated effusions can be broken down. Mechanical pleurodesis can be performed.
Thoracoscopy, whether medical or surgical, allows comprehensive assessment. Suspicious pleural nodules can be sampled. The extent of disease can be evaluated. For diagnostic uncertainty after thoracentesis, VATS often provides definitive answers.
The invasiveness is higher than percutaneous approaches. Recovery takes longer. But sometimes, nothing else will do.
Light’s Criteria for Differentiating Transudates from Exudates
Before choosing any treatment pathway, classification matters. Light’s criteria remain the gold standard for distinguishing transudative from exudative effusions. This is not academic exercise – it fundamentally directs management.
An exudate meets at least one of these criteria:
-
Pleural fluid protein divided by serum protein greater than 0.5
-
Pleural fluid LDH divided by serum LDH greater than 0.6
-
Pleural fluid LDH greater than two-thirds the upper limit of normal serum LDH
If none of these criteria are met, the effusion is transudative. Simple enough, right?
But here is where it gets interesting. Light’s criteria have excellent sensitivity for exudates but can misclassify transudates. Patients on diuretics may have concentrated pleural fluid that meets exudative criteria despite having heart failure. The serum-pleural fluid albumin gradient helps correct for this.
Diagnostic Evaluation and Investigations
Diagnosis is where pleural effusion management either succeeds or stumbles. The wrong diagnosis leads to wrong treatment. Wrong treatment leads to complications, delays, and poor outcomes.
Essential Pleural Fluid Analysis Parameters
Every thoracentesis should yield fluid sent for comprehensive analysis. The essential parameters include:
|
Parameter |
Clinical Significance |
|---|---|
|
Total protein |
Transudate vs exudate differentiation |
|
Lactate dehydrogenase (LDH) |
Light’s criteria component; elevated in malignancy, infection |
|
Glucose |
Low in empyema, rheumatoid, malignancy |
|
pH |
Complicated parapneumonic effusion indicator |
|
Cell count and differential |
Neutrophil predominance suggests acute process; lymphocyte predominance suggests TB or malignancy |
|
Cytology |
Malignant cell identification |
|
Adenosine deaminase (ADA) |
Tuberculosis indicator when greater than 40 U/L |
The biochemical approach outlined by PMC emphasises that Light’s criteria are pivotal in distinguishing effusion types by comparing pleural fluid and serum protein levels, while cytology and microbiological studies ascertain underlying causes.
In regions where tuberculosis is endemic, ADA becomes particularly valuable. A pleural fluid adenosine deaminase level greater than 40 U/L is indicative of tuberculosis effusion, according to guidelines from the Ministry of Health and Family Welfare.
Imaging Modalities for Initial Assessment
Chest radiography typically identifies the effusion initially. The classic meniscus sign, blunting of the costophrenic angle, and opacification of the hemithorax provide initial clues. Lateral decubitus films can demonstrate fluid layering and estimate volume.
Computed tomography offers superior detail. Pleural thickening, nodularity, loculations, and underlying parenchymal disease become visible. Contrast enhancement can suggest malignant involvement.
Ultrasound, already discussed for procedural guidance, also characterises effusions. Anechoic effusions are typically simple transudates. Complex septated effusions suggest exudative or infected processes. These findings guide initial management decisions.
Advanced Diagnostic Procedures for Uncertain Cases
Sometimes fluid analysis is inconclusive. Cytology negative. Cultures negative. Clinical suspicion high for malignancy or tuberculosis. What then?
Closed pleural biopsy can sample pleural tissue percutaneously. Yield improves with image guidance. However, thoracoscopy provides superior sensitivity for malignant pleural disease, allowing targeted biopsies of suspicious areas.
Medical thoracoscopy, performed under local anaesthesia and sedation, represents a middle ground between percutaneous approaches and VATS. Direct visualisation, biopsy capability, and the option for talc poudrage all become available.
Biomarkers and Laboratory Tests for Specific Conditions
Beyond standard parameters, specific biomarkers aid diagnosis:
-
Amylase – elevated in oesophageal rupture and pancreatitis
-
Triglycerides – greater than 110 mg/dL suggests chylothorax
-
Cholesterol – elevated in cholesterol effusion (pseudochylothorax)
-
NT-proBNP – helps identify cardiac causes when Light’s criteria misclassify transudates
-
Mesothelin – elevated in mesothelioma
The key is ordering appropriate tests based on clinical context. Blanket testing wastes resources. Targeted testing yields answers.
Treatment Selection Based on Pleural Effusion Type
Pleural effusion causes determine treatment. A transudate from heart failure requires completely different management than an empyema. Malignant effusions demand considerations beyond drainage. This is where understanding pleural effusion pathophysiology pays dividends.
Managing Malignant Pleural Effusions
Malignant pleural effusions present unique challenges. The effusion itself is often symptomatic, but treatment must balance palliation against disease trajectory and patient goals.
Options include:
-
Repeated thoracentesis – appropriate for patients with limited life expectancy and slow fluid reaccumulation
-
Indwelling pleural catheter – allows home management, avoids repeated hospital visits
-
Talc pleurodesis – preferred for patients with expandable lung and reasonable performance status
-
Combination approaches – IPC placement with subsequent pleurodesis through the catheter
The IPC versus pleurodesis debate continues. Both approaches have merit. IPC offers faster hospital discharge and home management. Pleurodesis offers the possibility of definitive fluid control without ongoing drainage. Patient preference, performance status, and lung expandability guide the choice.
The real change was the conversation in the clinic. It stopped being doctors dictating treatment and started being patients choosing between genuine options with meaningful differences. That shift matters.
Approach to Parapneumonic Effusions and Empyema
Parapneumonic effusions develop in association with pneumonia. Most resolve with appropriate antibiotic therapy. But some progress to complicated parapneumonic effusions or frank empyema, requiring drainage.
Risk stratification guides intervention:
|
Category |
Characteristics |
Management |
|---|---|---|
|
Simple parapneumonic |
Small, free-flowing, pH greater than 7.2, glucose greater than 40 mg/dL |
Antibiotics alone |
|
Complicated parapneumonic |
Loculated or pH less than 7.2 or glucose less than 40 mg/dL |
Chest tube drainage plus antibiotics |
|
Empyema |
Frank pus or positive Gram stain/culture |
Chest tube drainage, consider intrapleural fibrinolytics, may need surgery |
Delayed drainage of complicated parapneumonic effusions leads to worse outcomes. Loculations form. Fibrin deposition creates septations. What started as a simple drainage becomes a surgical problem. The lesson? Intervene early when markers suggest complicated effusion.
Intrapleural fibrinolytics (tissue plasminogen activator) combined with DNase can improve drainage in loculated collections. The MIST2 trial demonstrated this combination’s superiority over placebo in reducing surgical referrals.
Treatment of Transudative Effusions
Transudative effusions reflect systemic problems. Heart failure. Cirrhosis. Nephrotic syndrome. The effusion is a symptom, not the disease.
Management targets the underlying cause:
-
Heart failure effusions respond to diuresis and optimisation of cardiac function
-
Hepatic hydrothorax often requires salt restriction, diuretics, and potentially transjugular intrahepatic portosystemic shunt (TIPS)
-
Nephrotic syndrome effusions improve with treatment of proteinuria and fluid management
Drainage provides symptomatic relief but does not address the root problem. Repeated thoracentesis for transudative effusions without treating the underlying cause is futile. The fluid keeps coming back.
Sounds simple, right? Fix the heart failure, and the effusion resolves. In practice, optimising these complex patients is anything but straightforward.
Special Considerations for Non-expandable Lung
Non-expandable lung – sometimes called trapped lung or lung entrapment – presents a management dilemma. After drainage, the lung fails to expand against the chest wall. A persistent pleural space remains.
Pleurodesis cannot work here. Without apposition of visceral and parietal pleura, there is nothing to fuse together. Attempts at pleurodesis cause pain without benefit.
Pleural manometry during thoracentesis helps identify this condition. Excessive negative pressure development during drainage suggests non-expandable lung. The IPC becomes the preferred management option, allowing symptomatic drainage without attempting pleurodesis.
And yet, even here nuance matters. Some cases of apparent non-expansion improve with time and repeated drainage. The lung gradually re-expands as adhesions release. Patience sometimes pays off.
Conclusion
Effective pleural effusion management demands precision at every step. The days of draining fluid and waiting are over. Understanding pleural effusion causes and matching treatment to underlying pathophysiology produces better outcomes. Ultrasound guidance is mandatory, not optional. Light’s criteria distinguish transudates from exudates and direct subsequent workup. Malignant effusions require individualised approaches balancing palliation with patient preferences. Parapneumonic effusions need timely intervention before complications develop. Transudative effusions demand attention to the systemic disease creating them. The tools exist. The evidence supports their use. Pleural effusion management, done properly, transforms respiratory distress into breathable relief.
Frequently Asked Questions
What is the success rate of talc pleurodesis for malignant pleural effusion?
Talc pleurodesis achieves success in approximately 84.6% of cases within 30 days post-procedure. Success depends on several factors, including patient performance status, lung expandability, and underlying malignancy type. Patients with better functional status tend to have improved outcomes, while those with trapped lung are poor candidates regardless of the sclerosing agent used.
When should thoracentesis be performed urgently?
Urgent thoracentesis is indicated when patients present with significant respiratory distress from large effusions, when empyema is suspected (fever, systemic sepsis with pleural effusion), or when there is concern for haemothorax. Clinical deterioration despite supportive care also warrants urgent intervention. Stable patients with moderate effusions can generally undergo semi-elective procedures with proper imaging guidance.
How does pleural effusion pathophysiology differ between transudates and exudates?
Transudates result from imbalanced hydrostatic or oncotic pressures. Elevated capillary hydrostatic pressure (heart failure) or reduced oncotic pressure (hypoalbuminaemia) causes fluid to accumulate without pleural membrane involvement. Exudates result from increased pleural membrane permeability due to inflammation, infection, or malignancy. The pleura itself is diseased, allowing protein-rich fluid to accumulate. This fundamental pathophysiological difference explains why transudates respond to systemic treatment while exudates require local intervention.
What are the main causes of pleural effusion in adults?
Common pleural effusion causes include congestive heart failure (most common transudate), pneumonia with parapneumonic effusion, malignancy (lung cancer, breast cancer, lymphoma, mesothelioma), pulmonary embolism, cirrhosis with hepatic hydrothorax, tuberculosis, and connective tissue diseases such as rheumatoid arthritis and lupus. Post-surgical effusions and traumatic haemothorax also occur. The underlying cause determines both prognosis and management approach.
Which symptoms indicate complicated pleural effusion?
Key pleural effusion symptoms suggesting complicated disease include persistent fever despite antibiotics, systemic toxicity, pleuritic chest pain, and failure to improve with initial treatment. Laboratory red flags include pleural fluid pH less than 7.2, glucose less than 40 mg/dL, LDH greater than 1000 IU/L, and positive microbiological cultures. Imaging findings of loculation or pleural thickening also indicate complexity requiring escalated intervention.
What is the role of pleural manometry in management decisions?
Pleural manometry measures pressure changes during fluid drainage. Excessive negative pressure development (typically more negative than -20 cmH2O) during thoracentesis suggests non-expandable lung. This information guides treatment selection. Patients with non-expandable lung are poor candidates for pleurodesis because the visceral and parietal pleura cannot appose. Indwelling pleural catheters become the preferred option. Manometry also helps predict post-procedure expansion pneumothorax and guides safe drainage volumes.







 We do what's right for you...
We do what's right for you...



